Fintel reports that on July 25, 2023, Needham upgraded their outlook for Verrica Pharmaceuticals (NASDAQ:VRCA) from Hold to Buy .
Analyst Price Forecast Suggests 139.53% Upside
As of July 6, 2023, the average one-year price target for Verrica Pharmaceuticals is 12.24. The forecasts range from a low of 10.10 to a high of $15.75. The average price target represents an increase of 139.53% from its latest reported closing price of 5.11.
See our leaderboard of companies with the largest price target upside.
The projected annual revenue for Verrica Pharmaceuticals is 5MM, a decrease of 43.84%. The projected annual non-GAAP EPS is -0.93.
What is the Fund Sentiment?
There are 91 funds or institutions reporting positions in Verrica Pharmaceuticals. This is an increase of 18 owner(s) or 24.66% in the last quarter. Average portfolio weight of all funds dedicated to VRCA is 0.04%, an increase of 44.12%. Total shares owned by institutions increased in the last three months by 31.42% to 13,268K shares. 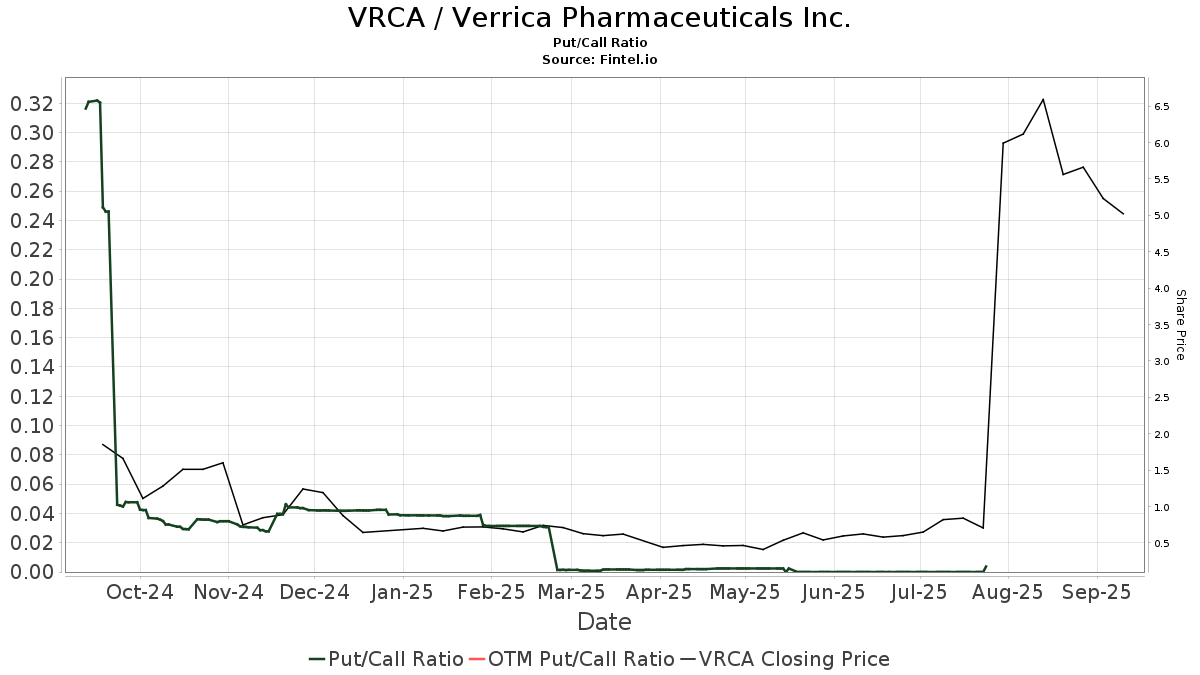 The put/call ratio of VRCA is 1.31, indicating a bearish outlook.
The put/call ratio of VRCA is 1.31, indicating a bearish outlook.
What are Other Shareholders Doing?
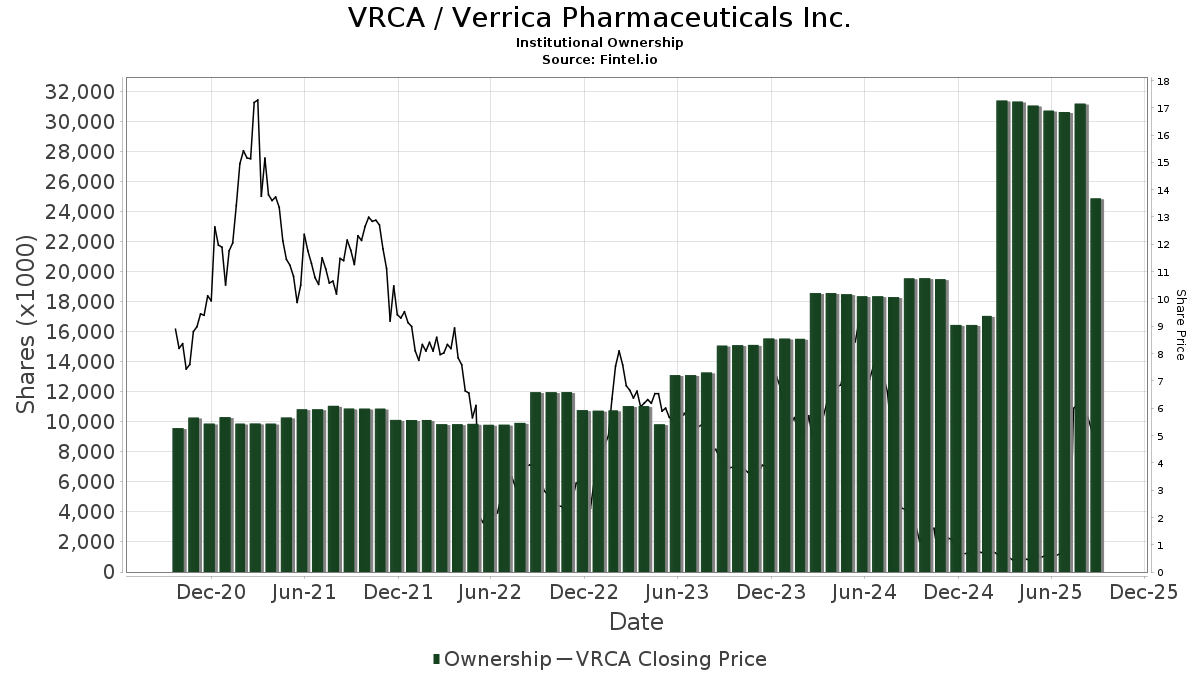
Perceptive Advisors holds 5,039K shares representing 12.04% ownership of the company. No change in the last quarter.
Avoro Capital Advisors holds 2,650K shares representing 6.33% ownership of the company.
FBIOX - Biotechnology Portfolio holds 1,452K shares representing 3.47% ownership of the company. In it's prior filing, the firm reported owning 1,598K shares, representing a decrease of 10.06%. The firm increased its portfolio allocation in VRCA by 133.19% over the last quarter.
VTSMX - Vanguard Total Stock Market Index Fund Investor Shares holds 815K shares representing 1.95% ownership of the company. No change in the last quarter.
VEXMX - Vanguard Extended Market Index Fund Investor Shares holds 267K shares representing 0.64% ownership of the company. In it's prior filing, the firm reported owning 176K shares, representing an increase of 33.85%. The firm increased its portfolio allocation in VRCA by 237.67% over the last quarter.
Verrica Pharmaceuticals Background Information
(This description is provided by the company.)
Verrica is a dermatology therapeutics company developing medications for skin diseases requiring medical interventions. Verrica's late-stage product candidate, VP-102, is in development to treat molluscum contagiosum (molluscum), common warts and external genital warts, three of the largest unmet needs in medical dermatology. Verrica is also developing VP-103, its second cantharidin-based product candidate, for the treatment of plantar warts. The Company has also entered a worldwide license agreement with Lytix Biopharma AS to develop and commercialize LTX-315 for dermatologic oncology conditions.
Additional reading:
- Verrica Pharmaceuticals Announces FDA Approval of YCANTH
- Disclaimer Certain information contained in this presentation and statements made orally during this presentation relates to or is based on studies, publications, surveys and other data obtained from third-party sources and Verrica’s own internal est
- Following FDA Approval of YCANTH™ for the Treatment of Molluscum Contagiosum Verrica Pharmaceuticals Enters into Non-Binding Term Sheet for up to $125 Million Debt Financing; Company to Host Conference Call and Webcast This Morning at 8:30 am ET – Te
- Verrica Pharmaceuticals Reports First Quarter 2023 Financial Results New Drug Application for YCANTH™ (VP-102) PDUFA Goal Date of July 23, 2023 - Potential to Become Only FDA-approved Therapy for Treatment of Molluscum Contagiosum Raised Gross Procee
- Verrica Pharmaceuticals Reports Fourth Quarter and Full-Year 2022 Financial Results New Drug Application for VP-102 assigned PDUFA goal date of July 23, 2023 VP-102 has potential to address the approximately six million patients in the U.S. with moll
This story originally appeared on Fintel.
The views and opinions expressed herein are the views and opinions of the author and do not necessarily reflect those of Nasdaq, Inc.


