Shares of Madrigal Pharmaceuticals MDGL lost 9.7% on Tuesday as the investors were not impressed by its preliminary fourth-quarter and full-year 2024 results. The company’s top line comprises only its product revenues. Madrigal’s commercial portfolio has one marketed drug, Rezdiffra (resmetirom), a once-daily, oral and liver-directed THR-β agonist.
Rezdiffra gained the FDA’s accelerated approval in March 2024 for adult patients with nonalcoholic steatohepatitis (NASH). Subsequently, the drug became the first and only FDA-approved therapy that targets the underlying causes of NASH.
In the past three months, Madrigal shares have gained 27.1% against the industry’s 11.7% decline.
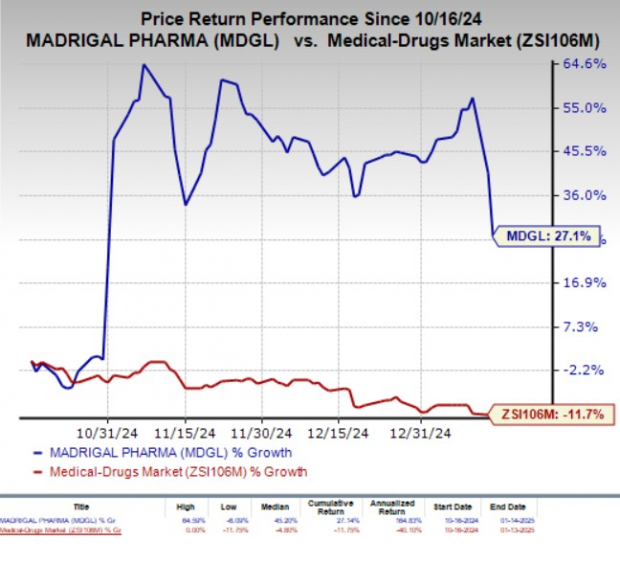
Image Source: Zacks Investment Research
MDGL’s Preliminary Q4 & FY24 Results
Madrigal reported preliminary fourth-quarter 2024 revenues from Rezdiffra sales in the $100-$103 million range.
For the full year, MDGL announced preliminary revenues in the $177-$180 million band from the drug’s sales since its launch in April 2024. Per the company, more than 11,800 patients are on Rezdiffra treatment as of year-end 2024.
Preliminary report suggests that Madrigal’s year-end 2024 cash, cash equivalents, restricted cash and marketable securities approximately amount to $931 million.
MDGL’s Pipeline Update
A regulatory filing, seeking approval for Rezdiffra for the same indication is currently under review in the EU. A final decision is expected in mid-2025. Subject to approval, Madrigal plans to initiate a country-by-country launch of the drug in the EU starting with Germany in the second half of 2025.
As the FDA approved Rezdiffra under the accelerated pathway, the continued approval will be based on promising long-term safety and efficacy data from the pivotal phase III MAESTRO-NASH biopsy study. This late-stage study, which provided the data for the drug's accelerated approval for NASH, is ongoing as an outcomes study. The goal is to generate confirmatory data that could verify the clinical benefits and support the full approval of Rezdiffra for the noncirrhotic NASH indication.
In addition to the MAESTRO-NASH study, a second phase III outcomes study is underway, evaluating the progression to liver decompensation events in patients with well-compensated NASH cirrhosis treated with Rezdiffra compared with placebo. This second outcomes study completed enrolling patients during the last reported quarter. Positive results from this study could make Rezdiffra the first approved treatment for patients with compensated cirrhosis, broadening its eligible patient group and potentially supporting full approval for noncirrhotic NASH.
These ongoing studies demonstrate Madrigal's commitment to establishing the drug as the standard-of-care treatment for NASH.
Madrigal Pharmaceuticals, Inc. Price and Consensus

Madrigal Pharmaceuticals, Inc. price-consensus-chart | Madrigal Pharmaceuticals, Inc. Quote
MDGL’s Zacks Rank & Stocks to Consider
Madrigal currently carries a Zacks Rank #3 (Hold).
Some better-ranked stocks from the sector are Castle Biosciences CSTL, CytomX Therapeutics CTMX and BioMarin Pharmaceutical BMRN, each sportinga Zacks Rank #1 (Strong Buy) at present. You can see the complete list of today’s Zacks #1 Rank stocks here.
In the past 30 days, Castle Biosciences’ earnings estimates for 2024 have remained constant at 34 cents per share. During the same timeframe, loss per share for 2025 has remained constant at $1.84. In the past three months, shares of Castle Biosciences have plunged 24.6%.
CSTL’s earnings beat estimates in each of the trailing four quarters, delivering an average surprise of 172.72%.
In the past 30 days, estimates for CytomX Therapeutics’ 2024 loss per share have remained constant at 5 cents. Estimates for 2025 loss per share have remained constant at 35 cents during the same timeframe. In the past three months, shares of CytomX Therapeutics have lost 28.7%.
CTMX’s earnings beat estimates in two of the trailing four quarters and missed the mark in the other two, delivering an average surprise of 115.70%.
In the past 30 days, estimates for BioMarin Pharmaceutical’s 2024 earnings per share have Improved from $3.28 to $3.29. Estimates for 2025 earnings per share have increased from $3.94 to $4.02 during the same timeframe. In the past three months, BioMarin Pharmaceutical shares have lost 9.1%.
BMRN’s earnings beat estimates in each of the trailing four quarters, delivering an average surprise of 28.7%.
Free Today: Profiting from The Future’s Brightest Energy Source
The demand for electricity is growing exponentially. At the same time, we’re working to reduce our dependence on fossil fuels like oil and natural gas. Nuclear energy is an ideal replacement.
Leaders from the US and 21 other countries recently committed to TRIPLING the world’s nuclear energy capacities. This aggressive transition could mean tremendous profits for nuclear-related stocks – and investors who get in on the action early enough.
Our urgent report, Atomic Opportunity: Nuclear Energy's Comeback, explores the key players and technologies driving this opportunity, including 3 standout stocks poised to benefit the most.
Download Atomic Opportunity: Nuclear Energy's Comeback free today.BioMarin Pharmaceutical Inc. (BMRN) : Free Stock Analysis Report
CytomX Therapeutics, Inc. (CTMX) : Free Stock Analysis Report
Madrigal Pharmaceuticals, Inc. (MDGL) : Free Stock Analysis Report
Castle Biosciences, Inc. (CSTL) : Free Stock Analysis Report
To read this article on Zacks.com click here.
The views and opinions expressed herein are the views and opinions of the author and do not necessarily reflect those of Nasdaq, Inc.


