Dr. Reddy’s Laboratories RDY and partner, Alvotech ALVO, announced the FDA’s acceptance of a regulatory filing, seeking the approval of AVT03, a proposed biosimilar of Amgen’s AMGN Prolia (denosumab) and Xgeva (denosumab), for review.
The biologic license application (BLA) was submitted under the 351(k) pathway, which is the FDA’s biosimilar application process. AVT03 is a human monoclonal antibody developed by Alvotech.
Amgen's Prolia treats osteoporosis in postmenopausal women at high risk of fractures, especially when other treatments are ineffective. AMGN’s Xgeva helps prevent fractures, spinal cord compression and the need for bone-related surgery or radiation in patients with multiple myeloma or bone metastases from solid tumors.
Both drugs play a crucial role in Amgen’s revenue stream. In 2024, Prolia generated $2.89 billion in sales, reflecting an 8% year-over-year increase. Meanwhile, Xgeva brought in $1.51 billion, marking 5% growth compared with 2023.
Year to date, Dr. Reddy’s shares have lost 15.5% compared with the industry’s 16.4% decline.
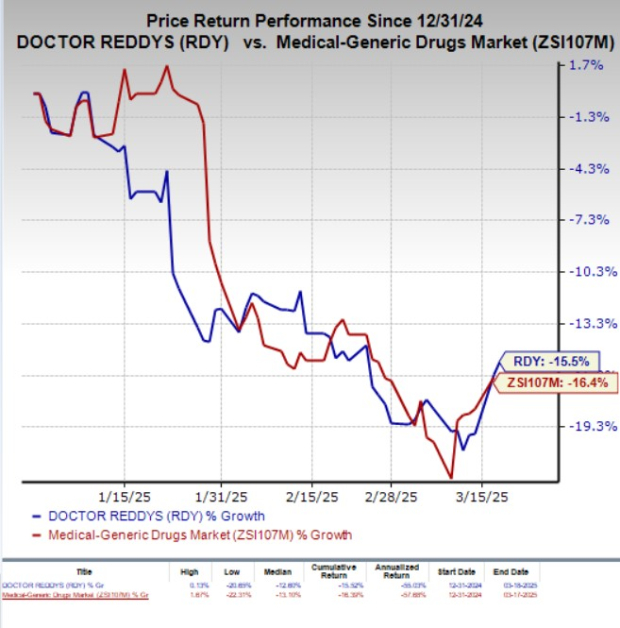
Image Source: Zacks Investment Research
Subject to approval, RDY/ALVO’s AVT03 could significantly enhance patient access to affordable treatment options for osteoporosis and other bone-related conditions. AVT03’s approval would align with broader efforts to expand the availability of high-quality biologic medicines. Given the high cost of branded drugs, a biosimilar alternative could provide substantial financial relief to patients and healthcare systems.
Additionally, with osteoporosis affecting millions across the United States, the introduction of AVT03 could improve treatment accessibility for a large patient population in need of effective bone health therapies.
RDY’s Ongoing Partnership With Alvotech for AVT03
Dr. Reddy’s and Alvotech signed a license and supply agreement for AVT03 in 2024, outlining their respective roles in bringing the biosimilar to market. Per the agreement, Alvotech is responsible for the development and manufacturing of AVT03, while Dr. Reddy’s is tasked with its registration and commercialization in key markets, including the United States.
Year to date, Alvotech shares have lost 14.3% against the industry’s 7.4% growth.
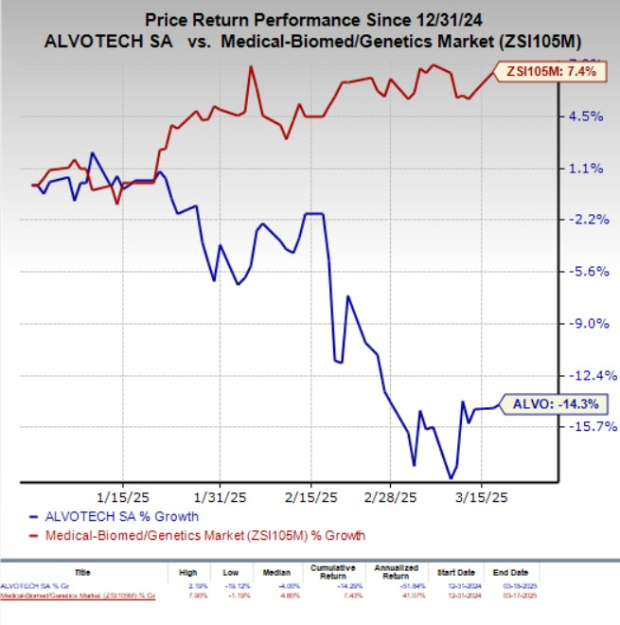
Image Source: Zacks Investment Research
Contingent upon the FDA’s approval, AVT03 is expected to be available in two formulations, a single-dose prefilled syringe containing 60 mg in a 1 mL solution and a single-dose vial with 120 mg/1.7 mL (70 mg/mL) solution. RDY and ALVO have clarified that AVT03 is an investigational product that has not been approved by any regulatory authority. Its biosimilarity has also not been established or claimed.
RDY & ALVO’s Zacks Rank and Stock to Consider
Dr. Reddy’s and Alvotech carry a Zacks Rank #3 (Hold) each at present.
A better-ranked stock from the sector is Gilead Sciences GILD, carrying a Zacks Rank #2 (Buy) at present. You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
In the past 30 days, Gilead Sciences’ earnings estimate for 2025 has improved from $7.80 to $7.87 per share. During the same timeframe, the estimate for earnings per share for 2026 has improved from $8.12 to $8.27. Year to date, shares of Gilead Sciences have gained 16.1%.
GILD’s earnings beat estimates in each of the trailing four quarters, delivering an average surprise of 19.47%.
7 Best Stocks for the Next 30 Days
Just released: Experts distill 7 elite stocks from the current list of 220 Zacks Rank #1 Strong Buys. They deem these tickers "Most Likely for Early Price Pops."
Since 1988, the full list has beaten the market more than 2X over with an average gain of +24.3% per year. So be sure to give these hand picked 7 your immediate attention.
See them now >>Dr. Reddy's Laboratories Ltd (RDY) : Free Stock Analysis Report
Amgen Inc. (AMGN) : Free Stock Analysis Report
Gilead Sciences, Inc. (GILD) : Free Stock Analysis Report
Alvotech (ALVO) : Free Stock Analysis Report
This article originally published on Zacks Investment Research (zacks.com).
The views and opinions expressed herein are the views and opinions of the author and do not necessarily reflect those of Nasdaq, Inc.


