Catalent’s CTLT shares surged 11.3% on Wednesday following the company’s promising update related to its pre-filled syringes. CTLT expects the booming demand for approved weight-loss drugs to keep the majority of its current and new production capacity booked until fiscal 2026.
The approval of GLP-1 class of drugs by the FDA for weight loss and diabetes in recent years has led to significant interest among patients, accelerating the demand for these drugs. Novo Nordisks’ NVO Wegovy and Ozempic, and Eli Lilly’s LLY Mounjaro and Zepbound are the FDA-approved drugs from the GLP-1 class. These drugs help people feel fuller for longer periods of time by affecting the hunger signals to the brain.
Lilly’s Mounjaro recorded $2.73 billion of sales in the first nine months of 2023, with $1.28 billion generated in the last reported quarter, reflecting strong uptake. The FDA approved Lilly’s second GLP-1 drug, Zepbound, earlier this month. Novo Nordisk’s sales within Diabetes and Obesity care increased 36%, led by 45% growth in sales of its GLP-1 drug, Wegovy. The drug recorded sales of more than $3 billion in the first nine months of 2023, reflecting nearly five-fold year-over-year growth. Ozempic sales grew more than 50% to $9.5 billion in the first nine months of 2023.
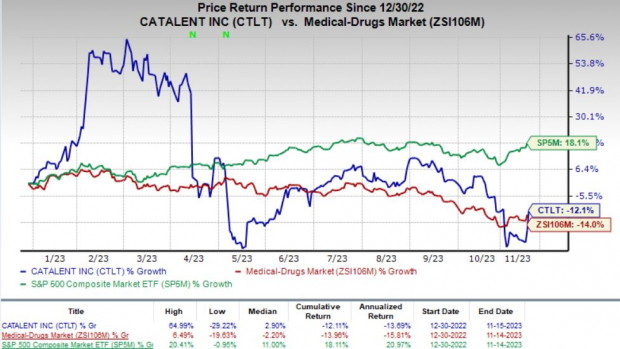
Image Source: Zacks Investment Research
Shares of Catalent have lost 12.1% year to date compared with the industry’s 14% decline. The S&P 500 Index has increased 18.1% in the same time frame.
Potential Growth of Pre-Filled Syringe
Catalent has already inked a contract drug manufacturer deal with Novo Nordisk to support the production of Wegovy by filling self-injection pens for the weight-loss drug. The strong demand trend for these self-administered GLP-1 class of drugs is likely to continue going forward, driving the demand for pre-filled syringes.
On its third-quarterearnings call Novo Nordisk raised its outlook for 2023 on the back of volume growth of GLP-1-based diabetes products. Novo Nordisk has high expectations from this segment, especially from Ozempic in the U.S. market and Wegovy in the obesity care market, post its relaunch. The anticipated rise in sales of Wegovy should boost Catalent’s pre-filled syringe order book.
CTLT is focusing on expanding its presence in the GLP-1 drug opportunity. The company is setting up new production capacity to support potential demand. It expects revenue contributions from GLP-1 drugs to exceed $500 million as expanded capacity gets operational. CTLT expects GLP-1 drugs to contribute $100 million in fiscal 2024.
On the back of strong GLP-1 potential, Catalent expects production capacity for pre-filled syringes to be booked out until fiscal 2026.
Apart from the GLP-1 drugs, the company’s focus on partnering with gene-therapy developers is also encouraging. Gene-therapy is an advanced class of treatment that has the potential to treat diseases with a single infusion. Catalent is already in partnership with leading gene-therapy developer, Sarepta Therapeutics SRPT, for supporting the manufacturing of the latter’s muscular dystrophy therapy, Elevidys. Sarepta is planning to scale up the manufacturing of Elevidys, which was approved in June 2023. Catalent expects a 65% increase in revenues from top customers, especially its biggest client, Sarepta.
These developments have fueled investors’ enthusiasm, thereby driving the stock price on Nov 15.
Catalent, Inc. Price

Catalent, Inc. price | Catalent, Inc. Quote
Zacks Rank
Catalent currently carries a Zacks Rank #4 (Sell).
You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
7 Best Stocks for the Next 30 Days
Just released: Experts distill 7 elite stocks from the current list of 220 Zacks Rank #1 Strong Buys. They deem these tickers "Most Likely for Early Price Pops."
Since 1988, the full list has beaten the market more than 2X over with an average gain of +24.0% per year. So be sure to give these hand-picked 7 your immediate attention.
See them now >>Novo Nordisk A/S (NVO) : Free Stock Analysis Report
Eli Lilly and Company (LLY) : Free Stock Analysis Report
Sarepta Therapeutics, Inc. (SRPT) : Free Stock Analysis Report
Catalent, Inc. (CTLT) : Free Stock Analysis Report
To read this article on Zacks.com click here.
The views and opinions expressed herein are the views and opinions of the author and do not necessarily reflect those of Nasdaq, Inc.


