Beam Therapeutics’ BEAM proprietary base-editing technology potentially enables the development of a differentiated class of precision genetic medicines that target a single base in the genome without making a double-stranded break in the DNA, thereby minimizing errors.
The company is pursuing two primary approaches depending on tissue type in vivo programs that use intravenously administered CRISPR as the therapy and ex vivo programs that use CRISPR to create the therapy using engineered human cells.
BEAM is evaluating its leading ex-vivo genome-editing candidate, BEAM-101, in the phase I/II BEACON study for sickle cell disease (SCD). It has successfully consented enough patients projected to both fill the sentinel cohort and initiate the expansion cohort of the early-stage study.
Apart from BEAM-101, the company is evaluating BEAM-201, a therapy designed to create allogeneic CD7, targeting CAR-T cells resistant to fratricide and immunosuppression. BEAM-201 is actively enrolling patients at multiple clinical sites, in a phase I/II study for treating patients with T-cell leukemia and T-cell lymphoblastic lymphoma, a severe disease affecting children and adults, and potentially other CD7+ malignancies.
The company also has two pre-clinical candidates —BEAM-301 and BEAM-302 — in its portfolio for genetic diseases.
BEAM has initiated IND-enabling studies for both candidates and plans to submit a regulatory application to begin clinical studies in the first half of 2024. Its shares have lost 43.9% year to date compared with the industry’s 18.3% decline.
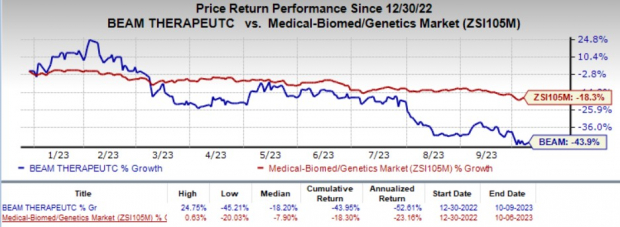
Image Source: Zacks Investment Research
While the company is yet to have an approved marketable product in its pipeline, its collaboration deals help access capabilities and resources to support the ongoing therapeutic programs.
BEAM collaborated with Pfizer PFE in 2021, focusing on using its gene-editing technology to create treatments for rare genetic diseases that affect the liver, muscles and the central nervous system. It is conducting the research work for three specific gene-editing programs. If these programs prove effective in clinical studies, Pfizer can choose to help develop and sell them globally, with both companies sharing the costs and profits.
A handful of other companies are also developing theirown in-vivo and ex-vivo gene-editing therapies.
CRISPR Therapeutics CRSP, in collaboration with Vertex Pharmaceuticals VRTX, is developing the investigational treatment exagamglogene autotemcel (exa-cel) for SCD and transfusion-dependent beta thalassemia (TDT). The companies have completed the BLA submissions with the FDA for exa-cel in SCD and TDT indications and a final decision regarding the same is expected by Dec 8, 2023, and Mar 30, 2024, respectively.
CRSP, along with VRTX, has also filed similar submissions on exa-cel in Europe and the United Kingdom.
Beam Therapeutics Inc. Price and Consensus

Beam Therapeutics Inc. price-consensus-chart | Beam Therapeutics Inc. Quote
Zacks Rank
BEAM currently carries a Zacks Rank #3 (Hold). You can see the complete list of today’s Zacks #1 Rank (Strong Buy) stocks here.
Pfizer Inc. (PFE) : Free Stock Analysis Report
Vertex Pharmaceuticals Incorporated (VRTX) : Free Stock Analysis Report
Beam Therapeutics Inc. (BEAM) : Free Stock Analysis Report
CRISPR Therapeutics AG (CRSP) : Free Stock Analysis Report
To read this article on Zacks.com click here.
The views and opinions expressed herein are the views and opinions of the author and do not necessarily reflect those of Nasdaq, Inc.


